Pipeline

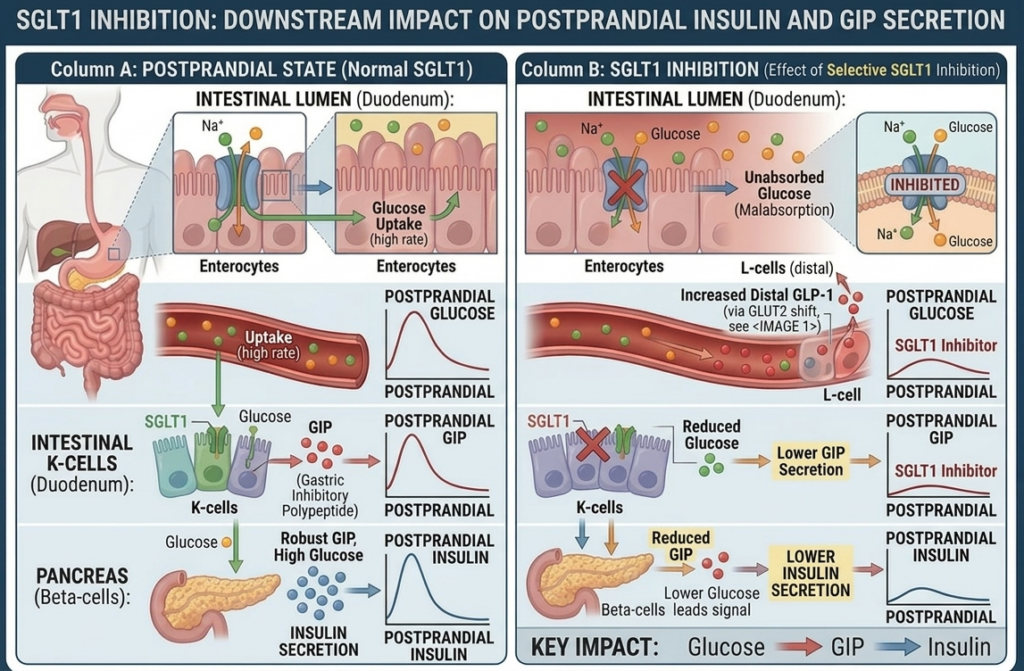
Mizagliflozin is an investigational first-in-class, orally administered, small molecule drug candidate that reduces postprandial glucose absorption, secretion of insulin, and secretion of gastric inhibitory peptide (GIP) by inhibiting sodium glucose transporter 1 (SGLT1) with a site of action in the intestinal lumen.
SGLT1 is the primary glucose and galactose transporter in the human small intestine and plays a secondary role in renal glucose reabsorption. Unlike sodium-glucose cotransporter 2, or SGLT2, which is localized mainly in the kidney, SGLT1 governs postprandial glucose absorption in the gut, making it a critical regulator of blood glucose excursions after meals. Pharmacological inhibition of SGLT1 reduces and delays the rate and extent of intestinal glucose uptake, blunting postprandial hyperglycemia and reducing insulin secretion as well as postprandial secretion of GIP. We believe this dual effect—attenuating postprandial glucose absorption while modulating incretin response—makes SGLT1 an attractive target for the treatment of metabolic diseases.
Mizagliflozin is in development by Vogenx for the treatment of PBH and Gastroparesis, debilitating diseases with high unmet medical need in underserved patient populations. Vogenx is also evaluating mizagliflozin as a treatment for rare metabolic disease GIP-dependent Cushing’s Syndrome. Mizagliflozin has been administered to over 500 subjects and has shown to be safe and well-tolerated in phase 1 an phase 2 clinical studies.
VGX-2857 is an investigational drug candidate under evaluation for the treatment of metabolic disease indications, including weight maintenance after substantial weigh loss.
