Post Bariatric Hypoglycemia (PBH)
PBH is a form of chronic postprandial hypoglycemia associated with bariatric surgeries. Bariatric surgery patients have been shown to overexpress SGLT1 in the intestinal lumen. Elevated levels of SGLT1 increases the quantity and rate of glucose absorption, leading to hyperinsulinemia. Glucose absorption also stimulates GIP secretion, that can further exacerbate hyperinsulinemia.
PBH is a relatively new disease with increasing awareness over the last several years. There are currently no FDA approved pharmacologic therapies for PBH. Current treatment options include dietary restrictions and the use of off-label drugs that have significant side effects with very limited and variable efficacy. The pathophysiology of PBH is complex and multifactorial, involving more mechanisms than solely changes in GLP-1 physiology. Recurring symptomatic and asymptomatic hypoglycemic events have been shown to be associated with long-term adverse comorbidities and are considered a marker of high-risk vulnerability for cardiovascular events, cognitive decline, dementia and higher mortality rates.
PBH patients have been shown to rapidly absorb glucose, causing increases in GIP secretion and abnormally elevated insulin (hyperinsulinemia). This hyperinsulinemia can result in low blood glucose and hypoglycemia events. During these hypoglycemic events, patients experience dangerously low blood sugar levels that can result in hypoglycemic symptoms such as confusion, weakness, dizziness, blurred vision, loss of consciousness and seizures.
Diagnostic criteria for PBH are evolving but generally include recurrent postprandial hypoglycemia (<54 mg/dL) occurring 1–3 hours after eating, with Whipple’s triad and exclusion of other causes. One particular challenge in developing diagnostic criteria is that studies have shown that many, if not most, hypoglycemic events in PBH patients are asymptomatic, even when blood glucose was less than 40 mg/dl. One key central and consistent condition appears to be inappropriately high secretion of postprandial insulin resulting in hypoglycemia.
PBH is generally believed to be an under reported spectrum disease with varying symptoms and levels of intensity ranging from mild to moderate and severe. Additionally, patients may be categorized as either symptomatic, where patients are aware of hypoglycemic events, and hypo-unaware, where patients do not have noticeable symptoms and are unaware of their hypoglycemic event. Therefore, categorizing PBH patients and even estimating the prevalence of PBH is particularly challenging. Recent studies generally suggest that between approximately 25% and 75% of bariatric surgery patients wearing continuous glucose monitors, or CGMs, exhibited PBH.
By inhibiting the SGLT1 transporter in the intestinal lumen of patients diagnosed with PBH in our clinical trials, mizagliflozin was shown to demonstrate statistically significant improvements in postprandial glucose peak, glucose nadir (the low point measured over a time course), insulin peak, GIP secretion, and measured hypoglycemic events.
Vogenx has initiated study start up activities for its phase 2b EMERGE clinical study of mizagliflozin in patients diagnosed with PBH. EMERGE is a US based, multicenter, randomized, double-blind, placebo-controlled, repeat dose parallel group study with a 21-day run in and 28-day treatment period. The primary endpoint will be a composite of the decrease of Level 3 and Level 2 hypoglycemic events (patient reported & blinded CGM). Patients and investigators interested to learn more about this and other upcoming studies should contact the Vogenx clinical team here.
Clinical Study VGX-001-012
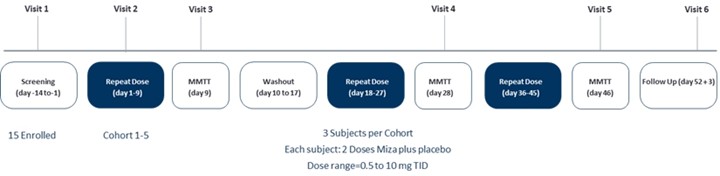
VGX-001-012 was a single-blind multicenter, Phase 2, randomized, placebo-controlled, ascending dose, crossover study. After enrollment, participants with PBH were randomly assigned to a treatment arm (5 cohorts, 3 participants per cohort; total of 15 participants). Each participant received placebo and 2 active doses in a crossover fashion for three 7-day treatment periods. Treatment with mizagliflozin resulted in robust improvement of glucose nadir, reduction of both peak glucose and insulin during the MMTT, and reduction in L3 and L2 events.
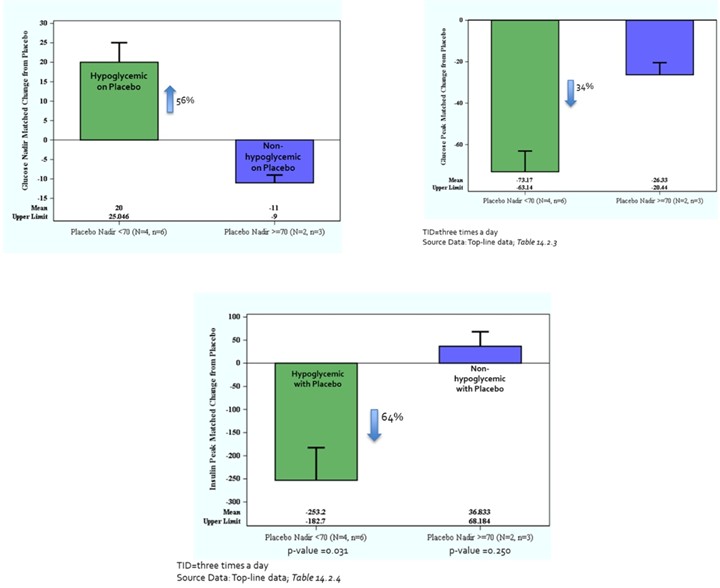
Compared with placebo, mizagliflozin 5 mg TID and 10 mg TID combined raised the glucose nadir by 56% (+20 mg/dL; p=0.028) and lowered the glucose peak by 34% (-73.2 mg/dL; p=0.031) and insulin peak by 64% (-253.2 μIU/mL; p=0.03) during the MMTT in a subgroup of patients that became hypoglycemic during the placebo mixed meal challenge.
Effect of Repeat Dose Mizagliflozin on Glucose Nadir, Peak Glucose, and Peak Insulin in PBH Patients after a Mixed Meal
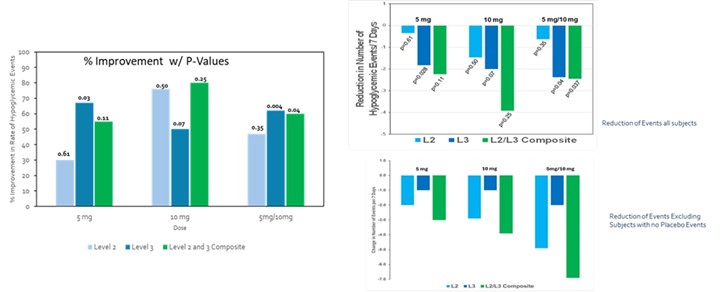
Significant reductions in rates of L3 hypoglycemia were observed (Figure below). With 5 mg TID, there was a 67% reduction of L3 hypoglycemia (p= 0.03) and with 10 mg TID, there was a 50% reduction of L3 hypoglycemia (p= 0.07) from placebo. There was a 30% and 76% reduction in L2 hypoglycemia with mizagliflozin 5 mg and 10 mg TID, respectively, although this was not clinically significant. When the composite endpoints of L2 plus L3 events were calculated, the 5 mg dose showed a 55 % decrease in events from placebo while the 10 mg dose showed a 80% decrease in events. When both dose groups were combined, the composite score was approximately 60% (p=0.04). The other bar graphs on the right show the reduction in the number of events either in all 5 mg and 10 mg subjects (top bar chart) or the reduction of the number of events excluding subjects that did not have Level 2 or Level 3 events on placebo (bottom bar chart). Mizagliflozin was well tolerated with no withdrawals or drug-related serious adverse events.
Clinical Study VGX-001-011
VGX-001-011 was a Phase 2 open label, randomized, single dose, crossover study. Nine participants with PBH were administered single doses of mizagliflozin and participants were randomly assigned to a treatment arm within a cohort receiving doses in a crossover fashion. The primary objectives were to evaluate the safety and tolerability of mizagliflozin in addition to the effects on postprandial glucose nadir in response to a mixed meal tolerance test (MMTT) compared to baseline.
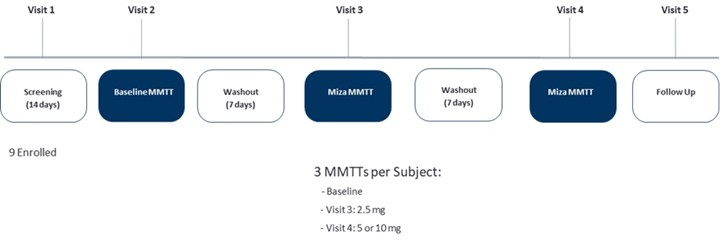
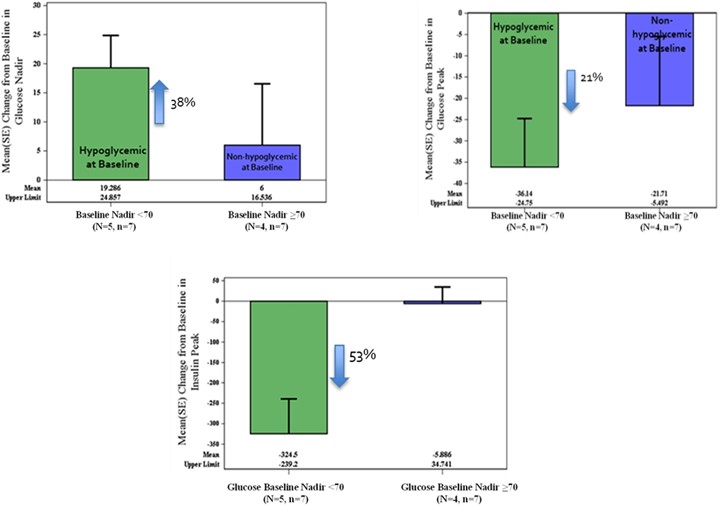
Compared with baseline, all doses of mizagliflozin (combined) raised the glucose nadir by 38% (p=0.03) and lowered the glucose peak by 21% (p=0.03) and insulin peak by 53% (P=0.016) in a subgroup of patients that became hypoglycemic during the baseline mixed meal challenge. Importantly, we did not observe significant changes in these parameters with PBH patients that did not go hypoglycemic in the baseline mixed meal challenge, suggesting mizagliflozin works only in the hypoglycemic situation. GIP area under the curve from 0-3 hours (AUC0-3h) was reduced by 504 pg hr/mL from baseline (p=0.048) in a subgroup analysis. Mizagliflozin was well tolerated and resulted in robust improvement of glucose nadir, reduction of both peak glucose and insulin, and reduction in postprandial GIP.
Effect of Single Dose Mizagliflozin on Glucose Nadir, Peak Glucose, and Peak Insulin in PBH Patients after a Mixed Meal